If you’ve ever walked into a tasting room, you’ve seen it: a room full of people rhythmically rotating their glasses like they’re trying to hypnotize the liquid inside. To a newcomer, it looks like a bit of “wine snobbery.” But in reality, swirling is a high-speed physics experiment that fundamentally changes the chemistry of what you’re about to drink.
Whether you’re a “cellar rat” on your first harvest or a seasoned pro, understanding the Science of the Swirl is the key to unlocking a wine’s full potential.
1. The Physics of Aromatics (Volatile Compounds)
The primary reason we swirl is aeration. Wine is packed with Volatile Organic Compounds (VOCs)—molecules responsible for those beautiful scents of cherry, oak, or passionfruit.
- Surface Area: When you swirl, you create a thin film of wine that coats the inside of the glass. This increases the surface area exposed to the air by up to 300%.
- Evaporation: That thin film on the glass wall warms up faster than the bulk liquid at the bottom. This “thermal nudge” increases the vapor pressure of the VOCs, launching them into the “headspace” (the empty area of the glass) so they can reach your nose.
2. The Chemistry of Oxygenation
Swirling isn’t just about smell; it’s about flavor and texture.
- Softening Tannins: For young, bold reds (like a Napa Cabernet), oxygen acts as a catalyst. It helps to “polymerize” or soften harsh tannins, making the wine feel silkier and less “grippy” on your palate.
- Removing “Off” Odors: Sometimes, a freshly opened bottle can have a slight “reductive” smell (like struck matches or boiled cabbage). A vigorous swirl acts like a breeze through a window, dispersing these unwanted gases and allowing the true fruit profile to surface.
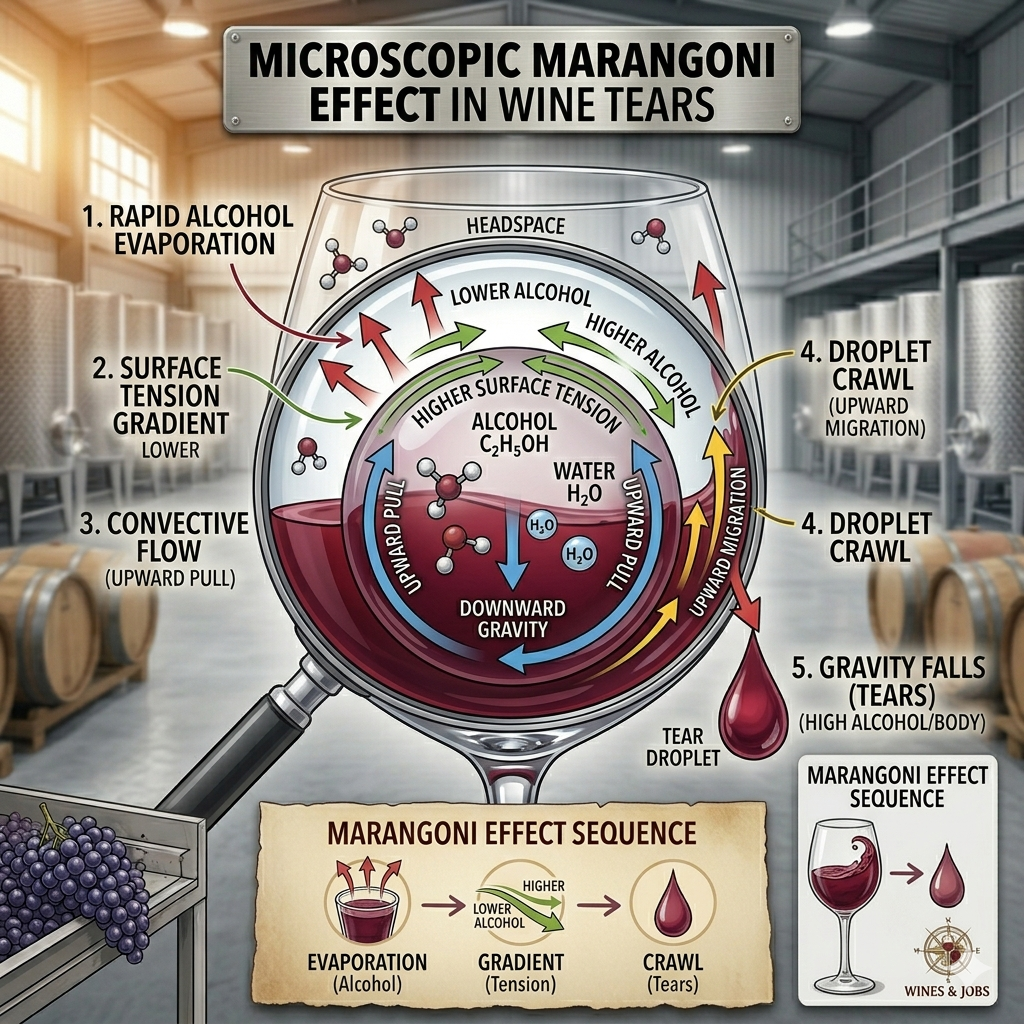
3. Reading the “Tears” (The Marangoni Effect)
After you stop swirling, you’ll notice clear droplets crawling back down the glass. These are often called “Legs” or “Tears.” Contrary to popular myth, legs do not indicate the quality of the wine. They are a result of the Marangoni Effect:
- Alcohol evaporates faster than water.
- This creates a surface tension gradient that pulls the liquid up the side of the glass.
- Eventually, gravity takes over, and the liquid falls back down in “tears.”
- The Insight: Thicker, slower legs generally indicate higher alcohol content or higher residual sugar, giving you a clue about the wine’s “body” before you even take a sip.
Swirling 101: How to Do It Like a Pro
If you’re worried about splashing wine on your shirt (or your neighbor), follow the Table-Top Method:
- The Pour: Never fill the glass more than one-third full. You need that headspace for the aromas to gather.
- The Grip: Place your thumb and forefinger at the base of the stem.
- The Motion: Keep the base firmly on the table and draw small, imaginary circles.
- The Sniff: Stop the motion, immediately put your nose deep into the glass, and take a short, sharp inhale.
🍷 Published on Wines and Jobs
Connecting the global wine community through insight, expertise, and opportunity.
In 2026, the industry is looking for professionals who don’t just “do” the ritual, but understand the science behind it. From sommeliers to laboratory technicians, the “physics of the glass” is a core part of the job.
- Find Your Next Role: Browse Sommelier & Lab Jobs
- Master the Ritual: Join our community for more technical deep dives at www.winesandjobs.com.
This video provides a fascinating visual explanation of the fluid dynamics behind wine legs and the Marangoni effect.
Cheers !!!

